Please check our recent publication in the Asian Journal of Organic Chemistry (IF 3.319), a collaboration with Professor Anthony Burke (Univ Évora) on the application of quantum mechanisms to understand the stereoselectivity of the transformation of a chiral glycolic acid to an isaurone.
Transformation of a Chiral Glycolic Acid to an Isoaurone: Stereochemical Assignment of a Benzilic Acid Rearrangment Product
Burke A. J., Teixeira C. S. S., Sousa S.F.
Asian Journal of Organic Chemistry
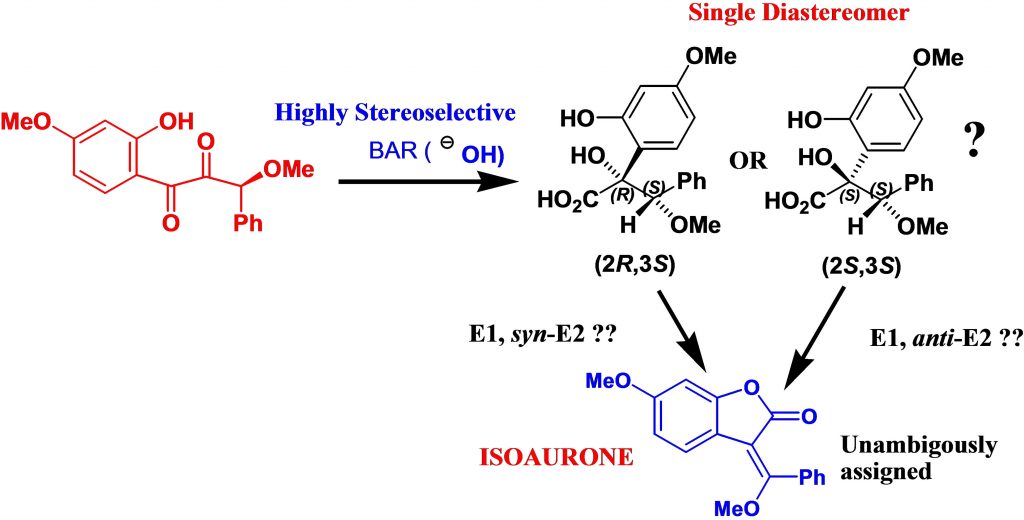
Abstract:The benzilic acid rearrangement (BAR) has been exploited with great effect over the last decades, used as the key step in many cases to afford complex target structures with interesting properties. Although the reaction is inherentely stereoselective in cyclic systems, the results achieved using acylic diketone systems can be less satisfactory. This report describes a highly stereoselective BAR in an acyclic system that gives a glycolic acid product which is then transformed via an elimination reaction to an unusual isoaurone product, in fact this latter product was the key to unravelling the stereochemistry of the glycolic acid product. The elimination reaction mechanism was studied using computational approaches.
