Please check our latest collaborative paper published in Bioorganic Chemistry, entitled “Benzo[a]phenoxazinium chlorides: Synthesis, antifungal activity, in silico studies and evaluation as fluorescent probes”
Benzo[a]phenoxazinium chlorides: Synthesis, antifungal activity, in silico studies and evaluation as fluorescent probes
Maria Inês P.S.Leitão, B.Rama Raju, Nuno M.F.S.A.Cerqueira, Maria JoãoSousa, M. Sameiro T. Gonçalves
DOI: 10.1016/j.bioorg.2020.103730 | Bioorganic Chemistry
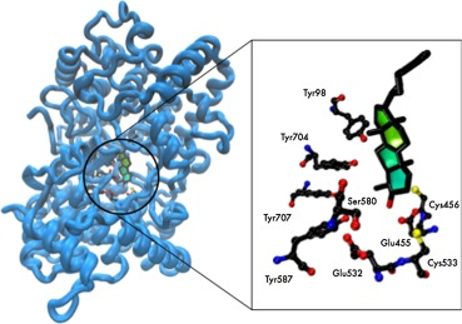
Four new benzo[a]phenoxazinium chlorides with combinations of chloride, ethyl ester and methyl as terminals of the amino substituents were synthesized. These compounds were characterized and their optical properties were studied in absolute dry ethanol and water. Their antiproliferative activity was tested against Saccharomyces cerevisiae in a broth microdilution assay, along with an array of 36 other benzo[a]phenoxazinium chlorides. Minimum Inhibitory Concentration (MIC) values between 1.56 and >200 µM were observed. Fluorescence microscopy studies, used to assess the intracellular distribution of the dyes, showed that these benzo[a]phenoxazinium chlorides function as efficient and site specific probes for the detection of the vacuole membrane. The added advantage of some of the compounds, that displayed the lower MIC values, was the simultaneous staining of both the vacuole membrane and the perinuclear membrane of endoplasmic reticulum (ER). Molecular docking studies were performed on the human membrane protein oxidosqualene cyclase (OSC), using the crystal structure available on PDB (code 1W6K). The results showed that these most active compounds accommodated better in the active sites of ER enzyme OSC suggesting this enzyme as a potential target. As a whole, the results demonstrate that the benzo[a]phenoxazinium chlorides are interesting alternatives to the available commercial dyes. Changes in the substituents of these compounds can tailor both their staining specificity and antimicrobial activity.
